Professor Jang Min Develops Ru-Doped Bismuth Titanate for Enhanced Ammonia Production
- admin
- 2024-12-09
- 1891
Professor Jang Min (Department of Environmental Engineering) and His Research Team Develop a Ru-Doped Bismuth Titanate Perovskite Oxide Photocatalyst
That
Enhances Ammonia Production Through Nitrogen Fixation
Dr. Thet Swe Zan, a postdoctoral researcher and
first author, along with Research Professor Jong Cho-Eun (corresponding
author), Professor Choi Eun-Ha (Director of PRBC), and Professor Yoon Yeo-Min
(Ewha Womans University), all from Professor Jang Min's research team
(Department of Environmental Engineering, Kwangwoon University), have
successfully developed a Ruthenium (Ru)-doped Bismuth Titanate photocatalyst
that significantly enhances ammonia production without requiring sacrificial
reagents.

Ammonia is a key industrial chemical primarily
used in fertilizer production and as an energy storage material that can be
converted into hydrogen. It is typically synthesized through the Haber-Bosch
process, which involves combining nitrogen and hydrogen under high pressure and
high temperature in the presence of a catalyst. However, the Haber-Bosch
process is highly energy-intensive and heavily reliant on fossil fuels to
produce the necessary hydrogen, resulting in significant greenhouse gas emissions.
With the global demand for ammonia rising exponentially, there is an urgent
need to develop more sustainable production methods.
Photocatalytic nitrogen fixation, which converts
atmospheric nitrogen and water into ammonia using sunlight as an energy source,
is an emerging technology with significant potential. While it is more
environmentally friendly compared to the conventional Haber-Bosch process,
substantial research and development are still required to optimize the
efficiency, scalability, and economic feasibility of photocatalytic ammonia
synthesis. Sacrificial reagents, such as methanol or ethanol, are effective in
increasing ammonia yield through photocatalytic nitrogen fixation but come with
the drawback of higher operating costs.
Bismuth titanate (Bi4Ti3O12)
has a bandgap structure suitable for photocatalytic nitrogen fixation to
produce ammonia. However, it has limitations due to restricted separation of
photoexcited electron-hole pairs, weak interactions with N₂, and a tendency to favor
hydrogen evolution reaction (HER), which hinders its efficiency. In response,
Professor Jang Min’s research team developed a Ruthenium (Ru)-doped Bi4Ti3O12
photocatalyst aimed at enhancing the interaction between N₂ molecules and active
sites on the photocatalyst surface to produce ammonia without the use of
sacrificial reagents. The ammonia production rate of the Ru-Bi4Ti3O12
photocatalyst was 3.2 times higher than that of the pure Bi4Ti3O12
photocatalyst. In the absence of sacrificial reagents, a novel result was
observed in which ammonia formation occurred through the oxidation of N₂ into NOx
species at the valence band of the photocatalyst, followed by the
photo-reduction of NOx species at the conduction band to produce ammonia. This
mechanism was further validated using in-situ SERS Raman and time-lapse FTIR
analyses, which demonstrated that Ru doping enhances the interaction between N₂ molecules and Bi sites,
facilitating N₂ photo-oxidation.
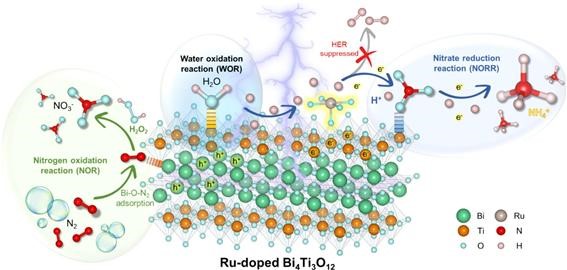
[Principle of Nitrogen Fixation and Ammonia
Production by Ru-Doped Bi4Ti3O12
Photocatalyst]
This study was published in the journal Applied
Surface Science (JCR Impact Factor 6.3, ranked first in the field of Materials
Science (Coatings and Films), JCR percentile: 97.83%). It was supported by
research funding from the National Research Foundation of Korea (NRF) under the
Ministry of Education (RS-2023-00240726, RS-2023-00282898, 2021R1A6A1A03038785,
and 2023R1A2C1003464).
https://www.sciencedirect.com/science/article/abs/pii/S0169433224026060
JENTL homepage: jentl.net
https://www.kw.ac.kr/ko/life/research.jsp?BoardMode=view&DUID=48544?